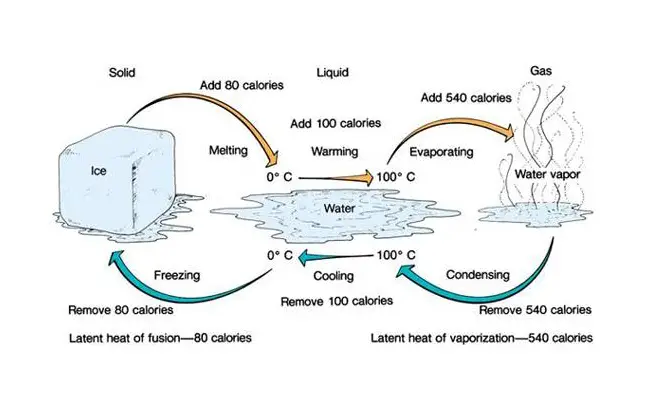
Latent heat refers to heat that is engrossed or freed an effect of a point change. No temperature is required during the change and therefore there is no shift in kinetic energy of particles within the matter. The freed energy is derived from the hidden energy deposited within the bonds inside the particles. Essentially, latent heat converts the state of the material from fluid to gas or from solid to fluid without changing temperature levels. For instance, a person sweat can exceed a liter in an hour under strained conditions of exercise or heat or untainted cocoa butter remains solid up to a 34℃ temperature where it melts pretty fast.
Uses of Latent Heat in Day to Day Life
Latent heat is used in various ways every day. It can be used to steam food in situations where vaporization of water is huge. Plates full of food absorb the heat from a hot steam. As the steam condenses at the plate’s base, large amounts of heat are released which in turn steam the food. Latent heat is also used to cool drinks. Adding ice or cold water to a cup of hot water speeds up the cooling process. As the ice melts, a large amount of latent heat is engrossed from a drink causing its temperature to drop. In the melting process, ice engrosses huge amounts of latent heat. When quenching a fire with boiling water, the water is quickly converted to steam which in turn absorbs higher amounts of latent heat from a fire. Latent heat also plays out during ice melting using salt. Salt has a higher latent heat of fusion than ice. When poured on a road that has a thick ice layer, salt requires a higher amount of heat energy to engross ice energy causing it to melt.