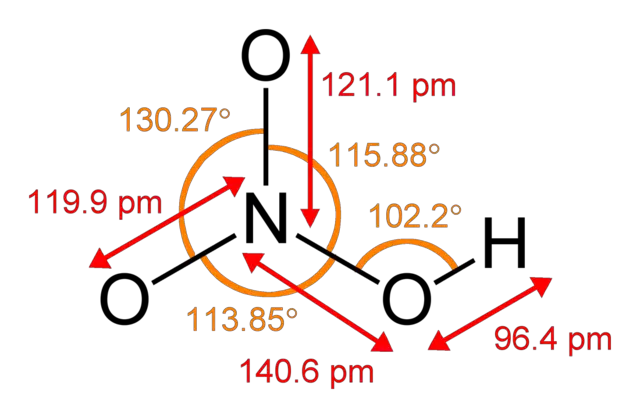
HN03 is the chemical symbol for the compound nitric acid. This acid is naturally clear and colorless but may become yellowish as it decomposes or comes in contact with water. Out of many inorganic compounds and acids, nitric acid renders itself useful to various industries including the explosives and fertilizer industry.
Most people may find nitric acid in 68% concentration. When this concentration is made to reach about 86%, it is then classified as fuming nitric acid. At this concentration level and even higher, nitric acid becomes useful in the manufacture of explosives and weapons. At high concentrations, nitric acid basically becomes the reagent for organic substances like the nitro group which are important component of explosives, especially those that react to changes in temperature and those that are shock-sensitive. The nitric acid used in this type of industry is considered stable enough for handling and safe use.
More stable concentrations of nitric acid may also be used as an oxidizing agent. Nitric acid easily reacts with water for example to speed up the oxidation process making it a good compound for this particular purpose. Diluted concentrations of nitric acid meanwhile may be used to help trace the presence of metals in a given solution. At 5% concentration and below, nitric acid in its purest and diluted form can help determine the presence of some metals. In the case of aesthetics, 10% concentration of nitric acid will help develop a rugged and aged look for some wood pieces like those from maple and fine trees. The wooden pieces in this case will have an oil wax finish from the grayish-gold color provide by the low concentration of nitric acid. The fertilizer industry also makes use of nitric acid because of its effect in providing essential nitrogen by reacting with other substances. When combined with water and alcohol, nitric acid also lends itself in the etching of metals and therefore helps to reveal the metal’s microstructure.