- The Idea of Rutherford
Ernest Rutherford, former head of the Cambridge University’s nuclear physics lab and the mentor of James Chadwick, was the first to introduce the idea that if protons had no mass, there could be something else in the atom that to yield mass but have no charges. He realized this when they were doing research and kept seeing that the number of protons was always less than the atomic mass. Rutherford called it a neutron, but he found no evidence at all.
- Chadwick’s Experiment
James Chadwick, a prisoner of war in World War I, and the student of Ernest Rutherford, provides evidence for Rutherford’s idea. As he saw the experiments of Frederic and Irene Joliot-Curie, he repeated their experiments with a goal of finding a particle with a mass but without a charge. He found out that the neutron really do exist with a mass of 0.1 percent more than the proton’s in 1932 and his experiments were a complete success.
- Possible Existence of Neutron
After his positive experiments, James Chadwick published his successful findings in his first paper entitled “Possible Existence of Neutron”. His paper was accepted and Werner Heisenburg showed the third particle of an atom, the neutron. This idea accelerated many discoveries involving the atom.
- Neutral Charge
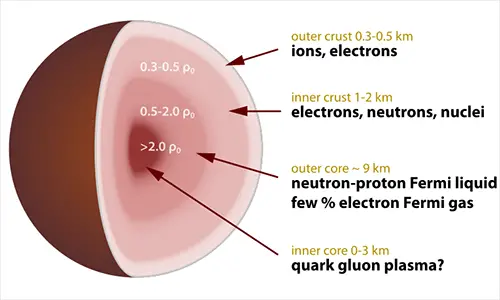
Unlike protons with a positive charge, and electrons with a negative charge, a neutron doesn’t have any charges, only mass. If you add more neutrons in an atom, you will create a radioactive charged atom.
- Radioactive Decay
A neutron and a proton can sometimes leave an atom during the process called Radioactive decay. An atom with a missing or an extra neutron is called an Isotope. An isotope contains the same number of protons and electrons. Even though a neutron has left an atom or has been added to another one, atom will still be the same element, it only creates a lighter or heavier version of that element.
- Neutron Computation
Computing the number of neutrons has a series of step. First is to find the mass number, which can be extracted by rounding off the atomic weight to the nearest whole number. After getting the mass number subtract it to the number of protons which is the same amount as the atomic number and you will get the number of neutrons in an atom.
- What is a Neutron?
A neutron is one of three particles found in an atom (protons, electrons and neutrons) located in the nucleus of an atom. Its mass was slightly higher than the proton and it has no charges. Neutron plays a major role in keeping the protons stable. Neutron was discovered in 1932 by James Chadwick.
- Neutron Therapy
This form of radiation therapy is used to treat cancerous tumors in any part of the body. It is the use of penetrating beams of ionizing radiation to treat this kind of disease. It damages the DNA strands to prevent the cancerous cells to infect other parts of the body. Proton and electron radiation are called low linear-energy-transfer (low LET) while the Neutron radiation is called high linear-energy transfer (high LET). Because of the neutron high-energy transfer the dose to kill cancer cells is only about 33% of the dose used in protons and electrons.
- Slow and Fast Neutrons
When a neutron has a series of collisions with different nuclei, the plural of nucleus, it will slow down. A neutron that slowed than is called “slow” and a neutron that has adopted its speed with its surrounding atoms is called “thermal”. Neutrons usually have 0.1 to 0.3 MeV before it is slowed down.







Leave a Reply